|
A common example of a tetrahedral molecule is CH 4 (methane). CO 3 (carbonate) is another example of a trigonal planar molecule. The bond angles in a trigonal planar molecule are 120 degrees. In molecular geometry, this is known as "trigonal planar". Also, for the structure to be correct, it. The total number of valence electrons between the two N atoms is 10e−. Since N is a member of the Group 5A (based on the periodic table), the number of electrons in its outermost shell must be 5. In order to come up with this answer, you first need to know the number of valence electrons for Nitrogen. Now, we have to identify the central atom in. Total number of valence electrons in N2H2 = 5*2 + 1*2 = 12. Hydrogen belongs to group 1 and has 1 valence electron. 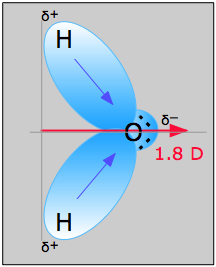
Nitrogen belongs to group 15 and has 5 valence electrons. In the case of N2H2, a single molecule has two atoms of nitrogen and two atoms of hydrogen.The common name for this molecule is acetylene.1 day ago transcribed image text: molecular geometry name of compound lewis structure bond angle(s) bond polarity (molecular structure/shape) n2 Expert Solution Trending now This is a popular solution! C2H2 has a straight-line molecular geometry consisting of a hydrogen atom bonded to a carbon atom, which is triple-bonded to a second carbon atom bonded to a second hydrogen atom. For example, the double-bond carbons in alkenes like C 2 H 4 are AX 3 E 0, but the bond angles are not all. When the substituent (X) atoms are not all the same, the geometry is still approxmiately valid, but the bond angles may be slightly different than the ones where all the outside atoms are the same. 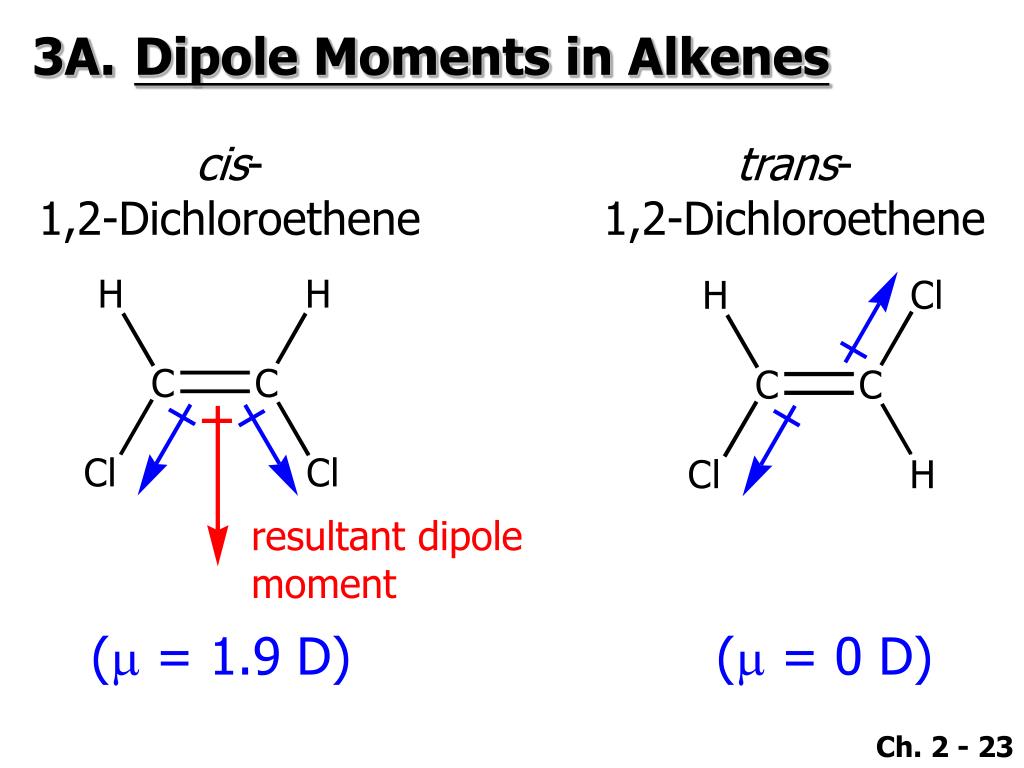
Both molecules have about the same shape and ONF is the heavier and larger molecule.† Geometry including lone pairs, shown in pale yellow ‡ Geometry excluding lone pairs. We clearly cannot attribute this difference between the two compounds to dispersion forces. 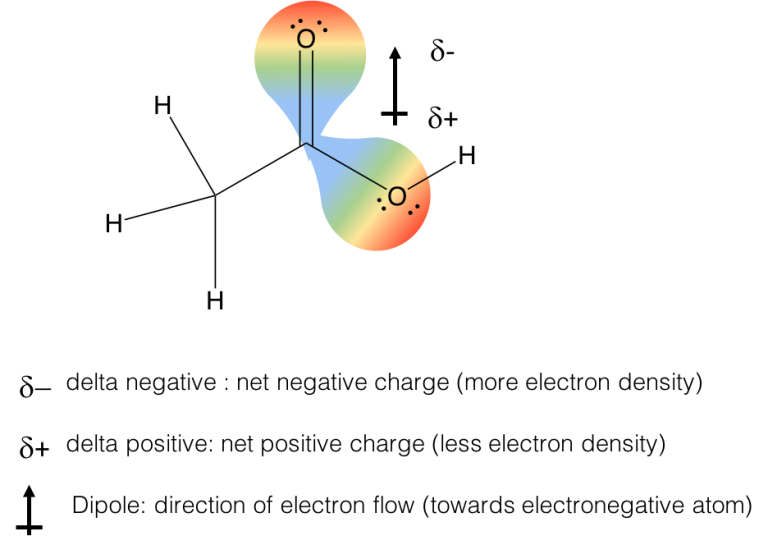
Water (H 2 O, molecular mass 18 amu) is a liquid, even though it has a lower molecular mass. The central Nitrogen atom has sp2 Hybridization with a bond angle of 118°.Nitrosyl fluoride (ONF, molecular mass 49 amu) is a gas at room temperature. There are two lone pairs of electrons in the structure. Each Nitrogen atom forms a pi bond with fluorine atoms. There is one double bond between both the Nitrogen atoms. N2F2 is made of two Nitrogen and two Fluorine atoms.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
